“We observed rapid and strong responses to this novel approach of treating patients with AFM13-NK, and we continue to evaluate the efficacy of this therapy for these hard-to-treat malignancies.” – Yago Nieto, M.D., Ph.D.
Imagine a breakthrough in cancer treatment that offers hope to patients who have exhausted all other options. That’s precisely what researchers have achieved with a groundbreaking Phase I trial involving natural killer (NK) cells complexed with a bispecific antibody called AFM13.
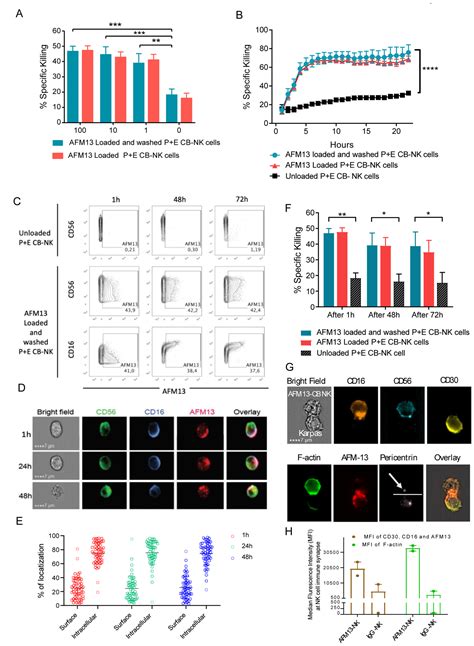
Published in Nature Medicine, the trial results unveiled an impressive 92.9% overall response rate and a complete response rate of 66.7% among 42 patients battling lymphoma. These individuals had been through rigorous prior treatments but found new possibilities with this innovative cell therapy.
Led by the esteemed Dr. Yago Nieto, the study not only demonstrated the effectiveness of AFM13-NK cells but also hinted at its potential application across various cancer types beyond lymphoma. Dr. Nieto highlighted, “These data lend to this approach being considered as a possible curative treatment for some patients and a bridge to a stem cell transplant for others.”
The magic of this approach lies in how Affimed’s AFM13 bispecific antibody binds to specific receptors on both NK cells and lymphoma cells, enabling targeted elimination of cancerous cells. This precise targeting mechanism enhances the therapy’s efficacy while minimizing harm to healthy tissues.
Dr. Katy Rezvani, another key figure behind this revolutionary technique, has been instrumental in developing impactful cell therapies at MD Anderson’s Institute for Cell Therapy Discovery & Innovation. Through her expertise, she continues to spearhead advancements that could transform cancer treatment paradigms.
The trial enrolled patients with CD30-positive Hodgkin lymphoma and T-cell lymphoma who were considered refractory to conventional treatments like brentuximab vedotin and anti-PD1 immune checkpoint inhibitors. Despite their challenging medical histories, participants experienced remarkable responses following treatment with AFM13-NK cells.
At follow-up assessments spanning up to two years post-treatment initiation, encouraging outcomes emerged – including high event-free survival rates and sustained complete remissions in several patients without requiring additional interventions. The therapy not only showcased promising efficacy but also boasted an excellent safety profile compared to traditional interventions.
Dr. Nieto emphasized the substantial implications of these findings: “Our trial showed the favorable safety profile and encouraging activity of AFM13-NK cells… not only [for] Hodgkin lymphoma but also supports future research into the clinical applications of NK cells with bispecific engagers.”
As we witness this transformative shift in cancer care through tailored cellular therapies like AFM13-NK cells, it becomes evident that hope is no longer just a distant beacon but a tangible reality for those fighting against formidable diseases like lymphoma.








Leave feedback about this