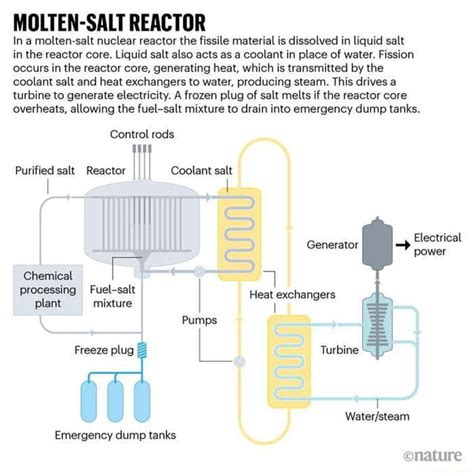
Imagine a world where nuclear energy production is not only safer but also more efficient and scalable. This dream is becoming a reality with the advent of molten salt reactors, an innovative technology that promises to revolutionize the way we harness nuclear power. But behind this groundbreaking advancement lies a crucial question: How do molten salts interact with other elements in a radiation environment?
Meet James Wishart, a distinguished chemist at Brookhaven Lab and the visionary leader of a pioneering research project delving into the intricate world of chromium chemistry in irradiated molten salts.
“Molten salt reactors are an emerging technology for safer, scalable nuclear energy production.”
In simple terms, molten salt reactors operate at higher temperatures than traditional water-cooled reactors by using a unique coolant composed of positively and negatively charged ions. To better understand these reactors’ long-term reliability, it’s essential to grasp how molten salts, especially chromium-laden alloys, behave under radiation.
“To assure the long-term reliability of these new reactors, we have to understand how molten salts interact with other elements in a radiation environment,”
explained Wishart.
Chromium, commonly found in metal alloys proposed for molten salt reactors, poses a significant challenge due to its propensity for corrosion. As chromium dissolves into the coolant, certain chemical forms can hasten corrosion processes, potentially jeopardizing the reactor’s structural integrity and performance.
“Chromium tends to be the easiest element to corrode from most alloys and will ultimately accumulate in the coolant of molten salt reactors,”
noted Wishart.
The key lies in understanding the oxidation states of chromium ions—specifically trivalent (Cr3+) and divalent (Cr2+). Trivalent chromium with three electron vacancies can accelerate corrosion, while divalent chromium with just two vacancies does not exhibit similar corrosive behavior. Hence, comprehending how these different forms react chemically within a radiation field is paramount.
Brookhaven Lab stands as a beacon of scientific exploration equipped with cutting-edge facilities capable of initiating and monitoring radiation-induced reactions on a minuscule time scale—from trillionths of seconds to minutes. Among these facilities are the Laser Electron Accelerator Facility and Van de Graaff accelerator housed within the Lab’s Chemistry Division.
Wishart and his team leveraged these advanced tools to scrutinize how Cr3+ and Cr2+ ions interacted with reactive species generated by radiation within molten salt environments. Their findings revealed that radiation tends to favor converting corrosive Cr3+ ions into less-corrosive Cr2+, shedding light on potential mitigation strategies for reactor degradation.
“Our analysis indicated that the net effect of radiation in the molten salt environment is to favor the conversion of corrosive Cr3+ to less-corrosive Cr2+,”
shared Wishart.
This groundbreaking research emerged from Brookhaven National Laboratory’s Molten Salts in Extreme Environments Energy Frontier Research Center—a DOE Office of Science initiative established in 2018 aimed at exploring fundamental properties and applications of molten salts within nuclear settings.
As we venture deeper into uncharted territories like molten salt reactors, studies like Wishart’s illuminate pathways towards safer and more efficient nuclear energy solutions while unraveling mysteries at the atomic level that shape our technological future.